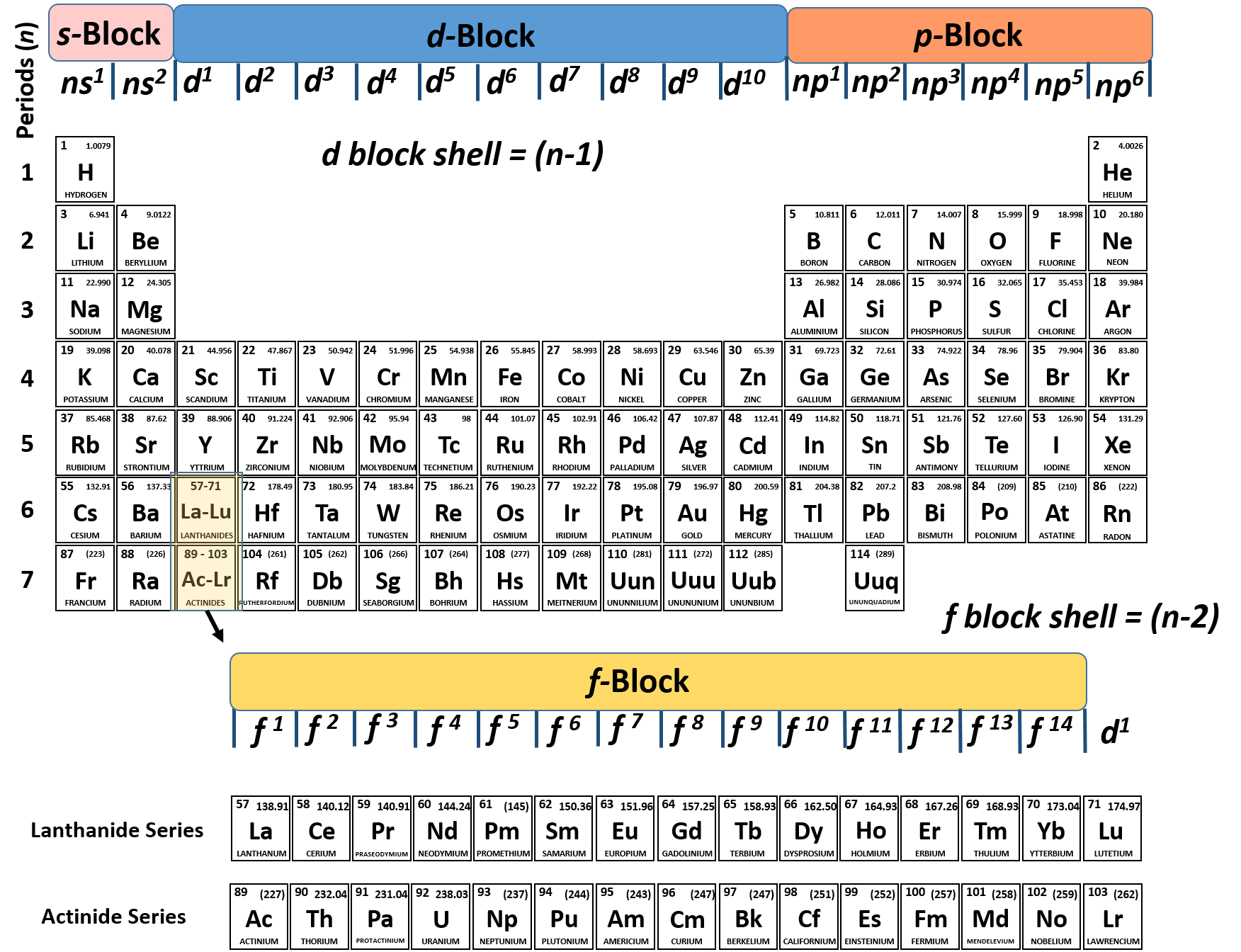
For more information on how to do this click here. You can also find the charge of an element by balancing a compound.All of the other elements have a charge when they have eight electrons all to themselves. That means that the charge of iron is 26-23= +3. Only the noble gases (the elements on the right-most column of the periodic table) have zero charge with filled valence octets. This particular can have different numbers of electrons but we’ll say this example has 23 electrons. Subtract the number of protons from the number of electron.Now let’s say you don’t have your handy periodic table with charges with you! There are multiple ways to find an elements charge. The relationship between ionic charge and the number of valence electrons for an element is an essential concept in chemistry. These trends are handy to memorize! Below you’ll find a full periodic table with the most common charges of every element! More Ways to Find a Charge Identify elements that will have the most similar properties to a given element. These isotopes are used as the charges on the periodic table for the elements. Explain the relationship between the chemical behavior of families in the periodic table and their valence electrons. Scientists then decipher the most common isotope. If for instance, one fluorine atom (9 protons) has 10 electrons giving it a -1 charge, but another fluorine atom has 9 electrons giving it a neutral charge, how do we know which charge to assume fluorine has? Well, these different variations of electron numbers are called isotopes. This element Valency PDF is a downloadable version of the Valences of the Elements table. This means it has one less electron than needed to complete the shell. However, atoms can often lose or gain electrons so it can be tricky to determine their charge. Excluding the transition metals and helium, any element on the periodic table will have the same number of valence electrons as the final digit of the group. The outer shell of a fluorine atom contains 7 electrons. 
Each element has a unique number of protons in the center of its atom, or its nucleus. Inorganic Chemistry: The Bond Valence Model (2nd ed.). A simpler version listing only the most common oxidation state charges is also available. What is a Charge?Ī charge on an atom comes from the difference between protons (positive charge) and electrons (negative charge) present in the atom. This information is available on a Color Periodic Table of the Elements or a Black and White version. The layout of the periodic table and where an element is located, can tell you a lot about its properties! In this article, you’ll learn how you can figure out what an element’s charge is by looking at where it’s placement on the periodic table! Here you’ll find a link to a downloadable periodic table with charges.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
